Congratulations to three bioengineers named Quad Fellows! Nya Domkam, BioE BS and MTM alumnus currently pursuing a PhD in Applied Sciences & Technology at Berkeley, Nandini Periyapalayam Sekar, first year PhD student, and Sharicka Zutshi, current BioE undergraduate planning to pursue graduate studies in medical diagnostics, have all been named to the 2023 class. The Quad Fellowship is an initiative of the governments of Australia, India, Japan, and the United States, designed to build ties among the next generation of scientists and technologists. The program sponsors 100 exceptional master’s and doctoral students in STEM to study in the United States.
mtm
Alison Burklund named to 30 Under 30
MEng alumna Alison Burklund, the cofounder and CTO of Nanopath, has been named to the Forbes 30 Under 30 list. Nanopath creates diagnostics that test pelvic and gynecologic infections during a single visit, so treatment can begin right away.
Decoding the Human Proteome With Glyphic Biotechnologies
Check out this feature on Bakar Labs startup Glyphic Biotechnologies – co-founded by MTM alumnus Josh Yang!
MTM alum named to 2022 30 Under 30
Master of Translational Medicine alumnus Steve Lee has been named to the Forbes 2022 30 Under 30 list in the social impact category! Steve and his brother Daniel founded Aura, an app that offers wellness and mental health coaching and resources.
2021 Best & Brightest MBAs- alumnus Joshua Young Yang
MTM alumnus Joshua Young Yang, co-founder of Nephrosant, is featured in Poets & Quants as one of the “2021 Best & Brightest MBAs.” Joshua is graduating from Stanford Business School and has founded another startup, Glyphic Biotechnologies.
Three BioE alumni in Forbes 30 Under 30!
Congratulations to three bioengineering alumni named to the 2021 Forbes 30 Under 30 list. Joshua Yang (MTM 2016) and Robert Chen (BS 2013) were named in Healthcare: Yang for his work on developing non-invasive tools to measure kidney injuries through startup company Nephrosant, and Chen for designing therapies for undernourished children, using links between certain gut bacteria and stunted growth. Joshua Nixon (BS 2016) and co-founder were named in the Social Impact list for founding Prime Roots, a company developing plant-based meat and seafood alternatives.
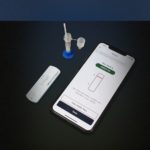
Gauss-Cellex rapid at-home Covid-19 antigen test
Gauss Surgical – a company launched by MTM alumnus Siddarth Satish – has partnered with Cellex to develop the first-ever rapid, at-home COVID-19 antigen test and mobile app.
Congratulations 2020 Graduates
Please join us in celebrating all of our 2020 graduates, who have showed so much dedication and perseverance getting through this wild year. We are so proud of all of you!
Bioengineers continuing the fight against COVID-19
After weeks of sheltering in place, Berkeley bioengineers are only increasing their efforts to mitigate the effects of COVID-19.