Session 2 Abstracts
Session 2 Abstracts
Session 2 Abstracts – Friday, June 22 – 1:30 PM
Biomaterials / Drug Delivery
Featured Speaker:
Controlled release of therapeutics and cells from hydrogel depots using light
Andrea Kasko, Assistant Professor, Department of Bioengineering, UC Los Angeles
Hydrogel scaffolds are used in biomedicine to study cell differentiation and tissue evolution, where it is critical to be able to deliver chemical cues in a spatially and temporally defined manner.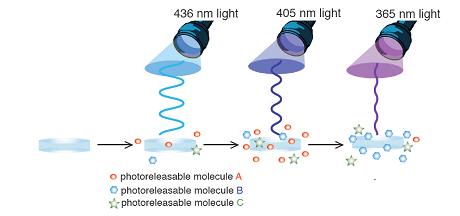 In order to recapture the complex cascades of signals and complex microenvironments found in nature, dynamic control over both the chemical and physical properties of a biomaterial is critical, and is typically achieved through controlled degradation and/or release. Hydrolytic and enzymatic degradation afford varying levels of temporal control but offer little spatial control. In contrast, photodegradation allows user-dictated real-time spatial and temporal control over the chemical and physical properties of a biomaterial, and allows for the controlled and triggerable release of therapeutic agents. We synthesized and characterized a series of macromers incorporating photodegradbale ortho-nitrobenzyl (o-NB) groups in the macromer backbone to allow physical patterning of the gels, and as a linker to a therapeutic agent to allow chemical patterning of the gels. We demonstrate the utility of these macromers and gels for the wavelength-biased release of therapeutic agents and whole cells. Through this approach, we are creating a sophisticated biomaterial platform that allows cell encapsulation along with real-time external control of the cellular environment in both time and space.
In order to recapture the complex cascades of signals and complex microenvironments found in nature, dynamic control over both the chemical and physical properties of a biomaterial is critical, and is typically achieved through controlled degradation and/or release. Hydrolytic and enzymatic degradation afford varying levels of temporal control but offer little spatial control. In contrast, photodegradation allows user-dictated real-time spatial and temporal control over the chemical and physical properties of a biomaterial, and allows for the controlled and triggerable release of therapeutic agents. We synthesized and characterized a series of macromers incorporating photodegradbale ortho-nitrobenzyl (o-NB) groups in the macromer backbone to allow physical patterning of the gels, and as a linker to a therapeutic agent to allow chemical patterning of the gels. We demonstrate the utility of these macromers and gels for the wavelength-biased release of therapeutic agents and whole cells. Through this approach, we are creating a sophisticated biomaterial platform that allows cell encapsulation along with real-time external control of the cellular environment in both time and space.
Selected Speakers:
Intestinal Permeability Increase During Ischemia Is Reduced By Enteral Serine Protease Inhibitors Angd and Cyclokapron
Angelina E. Altshuler, Alexander H. Penn, Jessica A. Yang, Itze Lamadrid, Diana Li, Stephanie Ma, Leena
Kurre, Lynn Han, and Geert W. Schmid-Schönbein
UC San Diego
Objective: During hypotension, the intestine becomes ischemic resulting in hemorrhagic necrosis, especially in the distal ileum, and inflammatory mediators are released into the peritoneal space, lymph, and circulation. Digestive serine proteases and lipases may contribute to the increased intestinal permeability and inhibitors to these enzymes serve to preserve the structure of the intestine in experimental shock. However, little is known about the effect of serine protease or lipase inhibition in the lumen of the intestine on intestinal permeability.
Methods: To produce complete intestinal ischemia, the proximal jejunum to the distal ileum was excised from male Wistar rats, flushed with saline, and sectioned into 8 equal segments. Segments were cannulated with tubing connectors, filled with 20 μg/ml fluorescein mixed with either saline, the serine protease inhibitors ANGD, aprotinin, or cyclokapron, and the lipase inhibitor orlistat, and sealed before placing in conical tubes filled with saline (N=6/group). The fluorescence intensity in the exterior fluid was determined at times 0, 30, 60, 90, and 120 minutes.
Results: Intestinal permeability increased during ischemia and reached higher levels in the distal ileum than the jejunum. ANGD and cyclokapron administration significantly reduced the rate of permeability compared to saline treatment, while orlistat and aprotinin, alone or in combination, were not effective.
Conclusion: During ischemia, permeability across the intestinal wall is increased in the distal ileum where hemorrhagic necrosis develops during ischemia. The rise of permeability was attenuated with ANGD and cyclokapron in the lumen of the intestine. These inhibitors may reduce the transport of inflammatory mediators from the lumen of an ischemic intestine. Supported by GM-85072.
Bioinspired Electrets for Solar Energy Conversion
Srigokul Upadhyayula,1 Robert Bonderer,1 Somaia Sarwat Sylvia,2 K M Masum Habib,2 M. Khalid Ashraf,2 Brent Millare,1 Amy Ferreira,1 Duoduo Bao,1 Bing Xia,3 Roger K. Lake,2 Valentine Vullev1
Department of Bioengineering, University of California, Riverside,1
Department of Electrical Engineering, University of California, Riverside.2
Department of Chemistry and Photonics Center, Boston University, Boston, Massachusetts.3
Protein-mediated electron-transfer processes sustain a broad range of redox functions in biological systems, such as respiration and photosynthesis. Therefore, such biological systems are inspirational models for materials for energy-conversion applications. This presentation will focus on bioinspired macromolecular electrets with rectifying long-range charge-transfer properties. We have demonstrated molecular designs based on anthranilamide derivatives, which similar to polypeptide α-helices, contain ordered arrangement of amide and hydrogen bonds, but in contrast to polypeptides and proteins, have an extended p-electronic conjugation for mediating efficient longrange charge transfer. Ab initio calculations, along with dielectric and NMR measurements, revealed that polyanthranilamides possess intrinsic dipole moments in the order of 3 D per residue. Furthermore, the macromolecular dipoles remove the degeneracy between the frontier orbitals of neighboring residues, causing splitting of ~0.2 eV for 10 residues that can readily result in rectification of vectorial charge hopping. Doping the macromolecules with electron withdrawing and electron donating groups did not only alter the bandgap and the energy levels of the frontier orbitals, but also modulated the intrinsic dipole moments, ranging from 0 to 5 D per residue. The design of the presented bioinspired electrets will prove instrumental for the development of materials with rectifying charge-transduction properties essential for solar-energy-conversion applications.
Windows to the Brain: Novel Concept for Providing Non-invasive, Chronic Access to Brain for Laser-Based Brain Tumor Diagnostics and Therapeutics
Yasaman Damestani, Guillermo Aguilar, Javier Garay, Devin Binder, Hyle Park, Masaru Rao
UC Riverside
Central Brain Tumor Registry of the U.S. estimates 66,290 new cases of primary and metastatic brain tumor in 2012. With current therapies including surgery, radiosurgery, and chemotherapy, the lifespan is limited to a median survival of 15 months while the 5-year survival is under 1%. Our goal is to develop a novel transparent polycrystalline Yttria-Stabilized-Zirconia (YSZ) cranial implant (“window”) that enables chronic, non-invasive delivery and/or collection of laser light into and from brain on demand. Our cranial implant thus allows for real-time and highly precise visualization and treatment of brain tumor without the need of future highly-invasive craniotomies or trepanation
procedures.The YSZ cranial implant is fabricated using current-activated powder assisted densification processing method. Waveguides have been written across millimeter-thick YSZ using femtosecond laser irradiation with ultralow energies. Cell growth and cytotoxicity assays have confirmed the biocompatibility of YSZ cranial implants with various surface properties. In animal study, a small part of skull was replaced with the implant to allow the delivery and/or collection of laser light precisely to affected areas within the brain which must be detected and treated chronically. The window was permanently covered with native scalp that could be rendered temporarily transparent in a minimally-invasive manner using percutaneous drug delivery of optical clearing agents (OCAs) with microneedles. Optical Coherence Tomography (OCT) images of YSZ implanted on the skull of mouse show that the transparent YSZ implant improves axial and lateral resolution and penetration depth into the brain in comparison with the skull of mouse and allows for visualization of brain tumor. The results from the present study suggest that YSZ implant in conjunction with OCA can be used as a minimally-invasive technique in delivering and/or collecting laser light to/from the brain for treatment and monitoring of brain tumor as well as other optical diagnostic and therapeutic applications.
Targeting Prostate Cancer Using Nanoparticles Decorated with the A11 Minibody
Kristine M. Mayle, Ricky Y.T. Chiu, Robert J. Lamm, Scott Knowles, Anna M. Wu, and Daniel T. Kamei
UC Los Angeles
Prostate cancer is the second leading cause of cancer-related death in American men. Current treatments are highly nonspecific, resulting in many negative side effects. In order to increase specificity, cancer therapeutics can be passively and actively targeted through encapsulation within nanocarriers and attachment of targeting ligands, respectively. In this study, we evaluated the potential use of the A11 minibody as a targeting ligand for systemic treatment of prostate cancer. The model drug delivery carrier was comprised of A11 and a layer of polyethylene glycol (PEG) conjugated to either poly(lactic-co-glycolic acid) (PLGA) or polystyrene nanoparticles. The size range of our nanoparticles allows passive targeting of delivery vehicles to tumors through the enhanced permeability and retention (EPR) effect. The PEG layer is used to improve the in vivo stability of the nanoparticles, and A11 serves the purpose of targeting the carrier to prostate stem cell antigen (PSCA), which is overexpressed on prostate cancer cells throughout all stages of the disease. Having been previously published as a positron emission tomography (PET) imaging agent, we would like to extend the use of A11 for drug targeting. The A11 minibody was successfully conjugated to PEGylated nanoparticles, as indicated by appropriate size increases. A11 was also found in vitro to significantly enhance the cellular uptake of PEGylated nanoparticles into PSCA transfected-22Rv1 prostate cancer cells (Figure 1). Moreover, we have been able to load small molecule anticancer drugs in our A11-conjugated PEGylated nanoparticles. Thus, A11 shows promise as a targeting ligand for drug delivery in the treatment of prostate cancer. Additional studies will be performed to elucidate the uptake mechanism of the A11-targeted PEGylated particles using endocytosis inhibitors. Furthermore, the ability of the A11 to enhance the chemotherapeutic effect of drug-loaded PEGylated nanoparticles, while reducing nonspecific toxicity, will be evaluated in the future.
Planar Microdevices Facilitate Transport of High Molecular Weight Therapeutics Across Retinal Epithelium
Jennifer S. Wade, Tejal A. Desai
UC San Francisco
PURPOSE
Due to the complications associated with intravitreal injection novel drug delivery technologies are desired. The
Desai Lab developed planar SU-8/PEGDMA microdevices using photolithography and coated them with the permeation enhancer Chitosan. These devices maximize contact surface area and provide consistent drug volumes. Using ARPE-19 coated inserts, the influence of geometry and surface coating on paracellular drug delivery to retinal tissue has been investigated.
METHODS
Device fabrication was achieved by a three-mask photolithography process. SU-8 and PEGDMA hydrogel solutions of FITC-Dextran (FD) or Lucentis were spun onto a silicon wafer. UV-light was used to cross-link the hydrogel in the device reservoir. Surface modification was conducted by deposition of a 1.6 % w/v Chitosan solution onto a drug-loaded wafer until dry film formation. Devices were deposited into the apical chamber of an ARPE-19 coated transwell insert. At various time points aliquots were removed from the basolateral chamber and analyzed for released drug concentration using a fluorimeter or spectrophotometer.
RESULTS
Microdevices with payloads of FD and Lucentis were repeatedly fabricated. Consistent elution of FD and Lucentis from devices was achieved. The quantity of FD transported across the ARPE-19 monolayer of cells using a microdevice was greater than the amount transported using a bolus deposition. The effect Chitosan coating has on the amount of drug transported is still being investigated and further optimization of the Chitosan coating process will clarify if the mucoadhesive inhibits drug elution.
CONCLUSION
A planar microdevice capable of housing therapeutics of varying molecular weight was developed. Preliminary data suggests this device enhances the transport of FD across an ARPE-19 in vitro retina model in comparison to bolus administration alone. Additional experiments to extend this work to Lucentis loaded microdevices are forthcoming.
Aptamer Functionalized Surface for Selective Capture of CD4 T cells
Qing Zhou, Ying Liu, Dong-Sik Shin
UC Davis
The quantification of leukocyte subsets in blood provides important diagnostic information. CD4 T-cells are central to mounting adaptive immune responses. Preferential killing of these cells by HIV results in drop of CD4 numbers and serves as the diagnostic marker of AIDS. Traditionally, monoclonal anti-CD4 antibodies are used to either label CD4 T-cells or to capture these cells on functionalized surfaces. Herein, we explored the use of CD4-specific RNA aptamer for affinity capture model T-cells on glass substrates. In the experiment, glass slides were first modified with acryl/amino silane. The surface was then incubated with Maleimide-PEGNHS linker, followed by thiolated CD4 aptamer. Subsequently, surfaces were passivated with BSA to minimize nonspecific adhesion. Ellipsometry was used to monitor assembly of different molecular layers and was used to demonstrate binding of recombinant CD4 antigen onto aptamer-modified surfaces. Adsorption of CD4 antigen on surfaces containing non-sense aptamers was not observed. In a proof of concept experiment, Molt-3 cells (CD4+) and Daudi B cells (CD4-) were labeled with cell tracker dyes, mixed at different ratios and infused into microfluidic devices with aptamer-modified surfaces. The results of cell capture experiments presented in Figure 1 demonstrate selective enrichment of CD4 expressing Molt-3 cells on aptamer modified surface. In these experiments Molt-3 cells were mixed with Bcells (Daudi) that do not express CD4. The data in Figure 1(e) shows Molt-3 capture purity of ~94% achieved based on cell mixtures where Molt-3 cells represented 15% and 25% of the cell population. In this study, we developed surfaces modified with CD4 aptamer and demonstrated high purity capture of CD4-expressing cells. The approach described here may be an alternative to antibody-based capture and quantification of CD4 T-cells in HIV/AIDS, with aptamers offering potential advantages of new cell detection schemes, chemical/thermal stability and regenerability.
Biomechanics / Mechanobiology
Featured Speaker:
More than a ‘feeling’: how cell mechanotransduction regulates stem cell lineage
Adam Engler, Assistant Professor, Department of Bioengineering, UC San Diego
Selected Speakers:
Load-mediated mechanical strains at a ligament-bone interface in vertebrates
A. T. Jang1, J. D. Lin1, D. Curtis1, S. Candell2, S. H. Lau2, K. Fahey2, M. I. Ryder3, S. P. Ho1
1Division of Biomaterials and Bioengineering, Department of Preventive and Restorative Dental Sciences, UCSF; 2Xradia Inc., Pleasanton, CA; 3Division of Periodontology, Department of Orofacial Sciences, UCSF
Aberrant loads can cause pathological deformations and over time can change the internal architecture of tissues and their interfaces. As a result, optimum function of an organ can be impaired if any of the interfaces or tissues within an organ adapt significantly. In this presentation, we will discuss how changes in functional loads can alter the overall displacement and the 3D association of the tooth with the alveolar socket. Shifts in biomechanical response of the organ and local strains within the bone-PDL-cementum complex harvested from rodents subjected to reduced functional loads will be discussed. Additionally, differential strains as a result of reduced functional loads, will be identified using an in situ mechanical device coupled to an X-ray microscope, and image correlation techniques. We theorize that changes in functional loads induce adaptations thereby altering the natural elastic gradients which further alter the loading, hence developing a positive feedback eventually leading to impaired function.
Shear stress modulates RAGE-mediated inflammation in a model of diabetes-induced metabolic stress
Justin Sherrod DeVerse, Keith A. Bailey, Kaleena N. Jackson, Anthony G. Passerini
UC Davis
Atherosclerosis occurs preferentially at sites of disturbed blood flow despite the influence of risk factors contributing to systemic inflammation. The receptor for advanced glycation endproducts (RAGE) is a prominent mediator of inflammation in diabetes that is upregulated in atherosclerotic plaques. Our goal was to elucidate a role for arterial hemodynamics in the regulation of RAGE expression and activity. Endothelial RAGE expression was elevated at sites of flow disturbance in the aortas of healthy swine. To demonstrate a direct role for physiological shear stress (SS) in modulating RAGE expression, human aortic endothelial cells (HAEC) were exposed to high SS (HSS, 15 dynes/cm2), which downregulated RAGE by 4-fold, or oscillatory SS (OSS, 0±5 dynes/cm2), which upregulated RAGE by 3-fold, compared to static culture at 4 hr. In a model of diabetes-induced metabolic stress, HAEC were chronically conditioned under high glucose (25 mM), then simultaneously stimulated with TNF-α (0.5 ng/ml) and the RAGE ligand high mobility group box 1 (HMGB1). A 50% increase in VCAM-1 expression over TNF-α was associated with increased cytoplasmic and mitochondrial ROS and NF-κB activity. This increase was RAGE-specific and NADPH oxidase-dependent. In activated HAEC, OSS amplified HMGB1-induced VCAM-1 (3-fold) and RAGE (1.6-fold) expression and proportionally enhanced monocyte adhesion to HAEC in a RAGE-dependent manner, while HSS mitigated these increases to the level of TNF-α alone. We demonstrate for the first time that SS plays a fundamental role in regulating RAGE expression and inflammatory responses in endothelium. These findings may provide mechanistic insight into how diabetes accelerates the non-random distribution of atherosclerosis in arteries.
Adult Cell Reprogramming and the Microenvironment
Timothy L. Downing, Jennifer Soto, Elaine Su, Constant Morez, Song Li
UC Berkeley
Many studies have outlined how a cell’s physical microenvironment can influence fate determination. Tight restriction on cell shape, for example, has the ability to curb mesenchymal stem cell (MSC) lineage commitment into adipocytes or osteoblasts via modulation of endogenous RhoA activity. This phenomenon was shown to be highly dependent upon actin-myosin generated cell tensions. Similarly, substrate rigidity has also been shown to direct MSC lineage specification through non-muscle myosin mediated substrate rigidity sensing mechanisms. Because most efforts to better understand the influence of matrix rigidity on cell fate determination focus solely on the differentiation and/or maintenance of select cell types, very little is known regarding how these matrix effects can influence cellular reprogramming and/or de-differentiation. To explore this new and emerging area of cell biomechanics we have utilized induced pluripotent stem (iPS) cell technology in conjunction with various bioengineered substrates. Primary fibroblasts were isolated from adult mice and expanded for up to three passages. Lentiviral vectors were then used to transduce adult mouse fibroblasts (aMFs) and force their expression of the four key reprogramming genes (Oct4, Sox2, Klf4, c-Myc). Following transduction, aMFs were seeded onto substrates of various rigidities and topographies. These substrates include polyacrylamide gels (rigidity ranging from 0.6 to 40 kPa) as well as random versus aligned nanofibrous membranes. Interestingly, our data largely suggest that 1) cytoskeletal and nucleoskeletal proteins play a critical role in the process of adult cell reprogramming and 2) by manipulating the physical microenvironment of adult fibroblasts we can alter cell reprogramming efficiencies (see Figure 1). Furthermore, these alterations in the physical microenvironment also yield changes at the epigenetic and transcriptional levels in aMFs. From this study, we gain a better understanding of the relationship between the physical microenvironment and cell reprogramming and, thus, gain insight into the mechanisms which govern iPS cell formation.
Mouse Embryonic Stem Cell-Derived Endothelial Cells Align on ‘Wrinkled’ Topography
Rachel Hatano1, Jesus Luna2, Drew Glaser2, Kevin Mercurio2, Valerie Leppert2, and Kara E. McCloskey2,3
1School of Natural Sciences, University of California, Merced
2Graduate Program in Biological Engineering and Small-scale Technologies, University of California, Merced
3School of Engineering, University of California, Merced
Emulating the micro- and nano-topography found in the extra cellular matrix of three-dimensional tissues is a vital technique to mimic and induce the cell physiology found in the native tissues. For example, the alignment of cardiac cells can be induced by distinct topographical signals, leading to enhanced cell-to- cell signaling and synchronous beating of the cell-tissue layer [1]. Utilizing Shrink-based technology founded by collaborator, Dr. Michelle Khine, we have also examined the ability for our mouse embryonic stem cell- derived endothelial cells (ESC-EC) to align on various microchip mold development systems. Briefly, we generated metal molds with ‘wrinkled’ topography using 1) 15nm and 2) 30nm of gold coating on the pre- shrunk polystryene (PS) sheets. After thermally shrinking, the mismatch between materials generated ‘wrinkles’ in the metal. Polydimethylsiloxane (PDMS) microchips were then generated from the wrinkled molds. We also generated 3) larger acetone-etched lines in the PS sheets, and 4) acetone-treated PS that generated ‘crazing’ topographical morphology. The ESC-EC were first characterized for endothelial purity, subphenotypes, and function by confocal microscopy, matrigel assays, and fluorescent activated cell scanning (FACS) analysis. The ESC-EC were then plated at a density of 20,000 cells/cm2 on the polydimethylsiloxane (PDMS) microchips made from the master wrinkled molds. After 1, 3, and 5 days of culture on the microchip, the cells were fixed, stained, and imaged using fluorescent microscopy. Based on actin staining and ImageJ analysis, the cells align from day 1, reach a maximum on day 3 (at subconfluence), but then decreased their alignment slightly on day 5 (at confluence). On day 3, 100 percent of cells aligned, defined as cells within 90°+30°, on the 15nm- and 30nm-coated mircochips. Whereas, 80% aligned on the acetone-etched surface and only 42% on the crazed surface, similar to the control (30%).
1. Luna, J.I., et al., Multi-scale Biomimetic Topography for the Alignment of Neonatal and Embryonic Stem Cell-derived Heart Cells. Tissue Eng Part C Methods, 2011.
Assessing Conservation of Human Heart Failure Mechanisms in Drosophila melanogaster
Gaurav Kaushik, Mayuko Nishimura, Rolf Bodmer, Anthony Cammarato, Adam J. Engler
UC San Diego
Due to its short lifespan, Drosophila melanogaster is a tractable system in which to study the effects of senescence on cardiac function. Female yellow-white (yw) flies experience a significant decrease in diastolic diameter between 1 and 5 weeks of age (~20%). An atomic force microscopy-based micromechanical analysis on the intact heart tubes of juvenile and geriatric flies revealed that their cardiomyocytes stiffen with age proximal to the intercalated discs (1.8 ± 0.13 to 3.8 ± 0.30 kPa). Another laboratory control strain, white (w1118), is significantly more arrhythmic but experiences less diastolic dysfunction with age (<10% decrease in diastolic diameter) and was found to have no change in stiffness with age (p > 0.10). DNA-microarrays have identified a host of mechanically-sensitive proteins to have significantly altered expression with age, particularly those involved with the cells’ ability to adhere to one another and their extracellular matrix. Quantitative polymerase chain reaction targeted against adherens junctions (AJ) molecules responsible for mechanical coupling of the myocytes reveals that they universally increase in expression with age in yellow- white, as observed in failing human hearts. Conversely, white flies experience persistent or diminished expression of AJ molecules with age. These first in situ mechanical analyses of living myocardium have resolved differences in passive cardiac mechanics due to age and suggest Drosophila as a novel platform to investigate the molecular mechanisms of human heart failure and their effects on cardiac cell function.
Inducing biofilm formation in Staphylococcus epidermidis clinical isolates using fluid flow in a microfluidic shear array
Westbrook M. Weaver, Vladana Milisavljevic, Jeff F. Miller, and Dino Di Carlo
UC Los Angeles
Staphyloccocus epidermidis is a significant cause of hospital-acquired infections, specifically blood stream infections (BSI) that result from the use of long-term central catheter lines. The ability to form biofilms is a key factor in the pathogenesis of these infections, with significant variation of biofilm phenotype amongst clinically isolated strains. The catheter lumen is a dynamic microenvironment inherently characterized by fluidic forces, however, little is understood regarding their effects on biofilm formation. Using a parallel microfluidic bioreactor shear array we show that fluid shear stress induces formation of significantly more biofilm than static conditions in a subset of clinically isolated strains, implicating catheter operation as a factor in bacterial pathogenesis.
Analysis of well-defined laboratory ATCC strains showed that the microfluidic biofilm assay could differentiate between biofilm+ (strain 35984) and biofilm– (strain 12228) genotypes with high confidence over a range of shears and culture times. We also found that the two clinical isolates A-10 and A-26, although both containing biofilm+ genotypes, display significantly different biofilm phenotypes under static conditions and flow.
Interestingly, the clinical isolate strain A-5, which is unable to form biofilms when grown in a 96 well-plate assay without flow, was induced to switch to a strong biofilm+ phenotype when grown under flow. Over a range of shear spanning two orders of magnitude, A-5 exhibited a biphasic dependence of biofilm formation on fluid flow, increasing biofilm secretion until mechanical forces overcame cell adhesion.
Our results show that certain strains significantly increase their pathogenic potential when exposed only to fluidic forces present in catheters under clinical operating conditions. These data lay the groundwork for further investigation into the molecular mechanisms involved in shear induced biofilm formation and, most importantly, warrant consideration of mechanical forces when designing catheter lumens and operational protocols, offering a potentially simple solution to avoid infection.
Synthetic Biology / Systems Biology / Computational Biology / Genomics
Featured Speaker:
How nature engineers enzymes for new functions
Patsy Babbitt, Professor and Vice Chair, Department of Bioengineering & Therapeutic Sciences, UCSF
Investigation of structure-function relationships in large and functionally diverse enzyme superfamilies suggests that many evolve multiple reactions using “privileged” scaffolds, structural templates whose active site architectures facilitate catalysis of common partial reactions or other chemical capabilities. Natural evolution has used such scaffolds to evolve many different reactions and reaction specificities consistent with these functional capabilities. A global view of these relationships reveals new insights about how new functions evolve and illustrates how an “underlying promiscuity” observed in these superfamilies can inform the functional assignment of proteins of unknown function and guide the choice of starting structures for enzyme engineering in the lab. These global studies also provide evidence for the convergent evolution of specific enzyme functions from different intermediate ancestors in the lineage of several of these superfamilies, providing a view of the multiple structural strategies that can be used to engineer enzyme reactions using these privileged scaffolds.
Selected Speakers:
Molecular analysis of the interaction between Staphylococcus aureus protein Sbi and immune system protein C3d
Wilson Rodriguez, Ronald D. Gorham Jr., Chris A. Kieslich, Aliana López de Victoria, Dimitrios Morikis
Department of Bioengineering, University of California Riverside
Protein-protein interactions form the basis of cellular communication, signaling, and regulation. These interactions are governed by non-covalent forces, originating from electrostatic and van der Waals interactions at atomic resolution. Electrostatic interactions are often crucial to protein recognition and binding, particularly for highly and oppositely charged proteins. We examined the interactions between secreted protein Sbi from antibiotic-resistant strains of S. aureus with host immune system protein C3d, using electrostatic clustering and free energy analysis. We used the computational framework called Analysis of Electrostatic Similarities Of Proteins (AESOP) to perform computational alanine scans and we generated a series of protein variants with perturbed electrostatic properties. To elucidate the effect of each mutation in association (defined as recognition plus binding) of Sbi with C3d, we performed electrostatic clustering and association free energy calculations. The analysis of our data revealed which charged amino acids are important for the formation of the C3d-Sbi complex, thus pointing out gain-of- association or loss-of-association mutations. This knowledge will form the basis for the design of proteins with tailored binding properties, and thus tailored biological function, by incorporating multiple critical mutations. This project will guide the development of novel therapeutics for treatment of infections by S. aureus.
Ultrasensitive Detection of Tuberculosis and Fungi Pathogens in Clinical Samples Using Modified Commercial ELISA with 4 Orders Higher Sensitivity
Abhinav P Acharya, Xuli Feng, Kousik Kundu, Ben Kline, Aaron Whiteley, Dan Portnoy, Niren Murthy
UC Berkeley
Introduction: Infectious diseases cause over 10 million deaths every year with a threat from certain pathogens to evolve into epidemic and pandemics. The areas of pathogen detection in the food and water sources and early detection of these pathogens in biological fluids have been studied extensively. However, there is a lack of technology that can accurately detect pathogens non invasively at an early stage of pathogenesis. In this report we demonstrate that the detection sensitivity of commercial ELISA kits can be increased by four to five orders of magnitude, using a new horse radish peroxidase (HRP) substrate composed of deuterocyanines coupled to a gold nanorod (D-Au). Results and Discussion: D-Au was generated by reducing gold conjugated cyanine dye via a linker. D-Au provided 20 picomolar sensitivity to H2O2 in Fenton’s reagent. Next, reactive oxygen species generated by HRP in the presence of H2O2 oxidized D-Au and provided 56 attomolar detection sensitivity of HRP as measured by a generic plate reader. HRP is the most commonly used reporter enzyme in ELISA, and hence D-Au is an excellent novel substrate for ELISA. We utilized D-Au in conjunction with commercially available ELISA kits to improve the detection sensitivity of cytokines such as IL-10, IL-12p40 and IL-1 by 4-5 orders of magnitude. Further, we demonstrate that D-Au based ELISAs are able to detect bacterial muscle infections in mice composed of a 1000 bacteria, via ELISA of the blood for leaked antigens. Next, infections induced by orally gavaged and intravenous injections of listeria monocytogenes could be detected after 3 days of infection in the stool samples of mice. Further, this is the first instance where an ELISA for detecting lipoarabionomannan an antigen produced by tuberculosis (TB) mycobacterium in saline and serum with high sensitivity was developed. This new ELISA technique is currently being utilized to evaluate antigen concentration in human bronchoalveolar lavage fluid for the presence of TB pathogen.
Mimicking the Second Secret of Life: Designing Artificial Cooperative Receptors
Anna J. Simon, Alexis Vallée-Bélisle, Francesco Ricci and Kevin W. Plaxco
UC Santa Barbara
The ability to precisely detect small changes in the concentration of specific molecules is key to many biosensing applications. The single site receptors typically employed for these technologies, however, suffer from a significant limitation: the physics of single-site binding fixes the dynamic range of such sensors, requiring an 81-fold increase in target molecule concentration to increase receptor occupancy from 10% to 90% and thus limiting their ability to precisely measure small changes in target concentration. The ability to detect small changes in the concentrations of specific molecules is, of course, also of significant importance to nature, and thus evolution has invented allosteric cooperativity, a mechanism by which the normally 81- fold dynamic range of single-site binding can be surmounted to produce a steeper, higher-sensitivity response, as is seen in hemoglobin’s binding to oxygen, bacterial chemotaxis receptors, and calmodulin signaling. Cooperativity arises when the first binding event on a multisite receptor triggers an increase in affinity for subsequent binding events, which in turn narrows the dynamic range of binding and steepening the input/output curve. We have adapted this same mechanism to improve the sensitivity of an artificial biosensor. Specifically, we have rationally re-designed molecular beacons, a widely employed optical and electrochemical sensor for the detection of DNA, to render it cooperative, thus significantly improving its ability to measure small changes in target concentration. To date we have developed 2-site molecular beacon architectures that reduce the dynamic range from 81-fold to within error of the theoretical limit of 9- fold and a three site architecture that reduces the dynamic range to ~7-fold. These architectures and variants of them should be easily adaptable to other nucleic-acid-based receptors and employed to implement highly sensitive input−output response in binding-activated biomaterials, nano-machines, drug-release devices, or synthetic biology systems.
Search of optimal drug combinations using an engineering feedback control scheme
Xianting Ding, David Jesse Sanchez, Arash Shahangian, Genhong Cheng and Chih-Ming Ho
UC Los Angeles
Herpes Simplex Virus 1 is one of the most pervasive infections in the world, causing genital, skin, and eye infection in millions of people around the world. Better and more effective methods of control are globally required in the face of drug resistance and the increased spread of this infection. Successful treatments of HSV with drug combinations have been reported. However, to determine an optimal drug cocktail is generally pricy, time consuming and labor intensive. The present study was undertaken to rapidly screen for combinations of IFN-α, IFN-β, IFN-γ, Ribavirin, Acyclovir and TNF-α against HSV-1 replication in vitro through feedback system control (FSC). We first focused on the ultimate goal of ablation of HSV replication by modifying the cellular state with drug combinations against viral replication. Using FCS we determined several combinations of FDA drugs that when added to infected cells could ablate virus growth with combined lower concentrations of drugs and better efficacy of treatment. We noticed that Ribavirin was a potent anti-viral drug, dominating the drug combinations. Ribavirin has been shown to cause anemia, and is teratogenic in some animal tests. We created a system to penalize against Ribavirin during drug combination screening using user-defined variables. Excitingly, we found that in fact we could determine new drug combinations without Ribavirin that were as affective. In both cases, optimal drug combinations were identified by testing less than 1% of total possible drug combinations, showing the merit of FSC on HSV-1 inhibition system.
Genetically engineering cellular mechanobiology
Joanna L. MacKay, Albert J. Keung, Sanjay Kumar
UC Berkeley
The physical properties of cells such as cell shape, size, and stiffness have recently been recognized as important regulators of cell behavior. For example, cells that are confined to a small area grow slowly and apoptose more frequently than cells grown in a large area (Chen et al., 1997 Science 276: 1425-1428), and cells that are polarized can migrate faster (Doyle et al., 2009 J Cell Biol. 184: 481- 490). As a novel strategy for controlling cell behavior, we have created cell lines in which we can directly tune the physical properties of cells. Specifically, we have introduced mutant versions of two intracellular mechanotransductive signaling proteins: RhoA GTPase and myosin light chain kinase, under a repressible promoter into glioblastoma cells using viral gene delivery. By varying the expression of either protein through controlled dosages of tetracycline, we demonstrate that we can modulate cytoskeletal architecture, traction force generation, cellular stiffness, and migration speed in a graded and dynamic fashion. We believe that this strategy will serve as a valuable tool for developing quantitative relationships between intracellular signaling pathways, cellular physical properties, and complex cell behaviors. Furthermore, such precise control over cell behavior could allow us to directly control how cells physically interact with their environment in tissue engineering applications. MacKay et al., 2012 Biophys J. 102:434-442
Dissecting a complex chemical stress: chemogenomic profiling of Zymomonas mobilis in plant hydrolysates and 37 components
Jeffrey M. Skerker1,2,3, Dacia Leon1,4, Morgan Price3, Jordan Mar1,4, Dan Tarjan1,4, Jason Baumohl3, Stefan Bauer1, Ana Ibanez1, Valerie Mitchell1, Cindy Wu4, Ping Hu4, Terry Hazen4, Adam P. Arkin1,2,3
1Energy Biosciences Institute, University of California, Berkeley
2Department of Bioengineering, University of California, Berkeley
3Physical Biosciences Division, LBNL 4Earth Sciences Division, LBNL
Plant hydrolysates are complex chemical mixtures containing fermentable sugars in addition to a wide variety of organic compounds, derived from sugar and lignin degradation, that are inhibitors of microbial growth and fermentation. The inefficient conversion of sugar to ethanol in these hydrolysates is a major hurdle to overcome in the realization of an industrial-scale cellulosic biofuel process. Here, we use pooled fitness assays and chemogenomics to uncover the genetic basis of hydrolysate tolerance in both Zymomonas mobilis and Saccharomyces cerevisiae. We first identified 49 genes in Z. mobilis and 28 genes in S. cerevisiae that are important for growth in miscanthus and switchgrass plant hydrolysates. By systematic overexpression of tolerance genes in Z. mobilis, we then identified a gene of unknown function that improves specific ethanol productivity 2.4-fold in the presence of miscanthus hydrolysate. To further understand how Z. mobilis responds to the complex mixture of chemicals present in plant hydrolysates, we analyzed our hydrolysates using GC-MS and LC-RID and identified 37 potential inhibitors. We obtained fitness profiles for each of these 37 compounds, and used this data to model hydrolysate fitness as a linear combination of its components. Of the 37 components in our model, 18 were sufficient to explain hydrolysate fitness. Synthetic hydrolysate mixtures containing all 37 compounds were not sufficient to explain the fitness profile of our real hydrolysates. Based on outliers in our synthetic hydrolysate data and linear model, we expanded our fitness dataset to include 11 other compounds or stress conditions that might explain the function of these outliers. Addition of these new conditions to our linear model, led to the identification of a previously unknown component of our miscanthus hydrolysate. Our work provides a general strategy to dissect how microbes respond to a complex chemical stress, and will enable future strain engineering efforts in both Z. mobilis and S. cerevisiae for improved biofuel productivity in plant hydrolysates.

