Posts Tagged ‘covid’
Using two CRISPR enzymes, a COVID diagnostic in only 20 minutes
Patrick Hsu, Liana Lareau and Daniel Fletcher have collaborated on a new rapid COVID test that rivals the sensitivity of the gold-standard qRT-PCR testing, with results in less than an hour.
Read MoreFDA authorizes Lucira home COVID-19 test
The FDA has authorized the first single-use, PCR-quality, over the counter COVID-19 test kit, made by alumni startup Lucira!
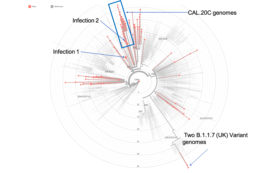
Read MoreLareau helps detect more infectious COVID variant at Berkeley
Professor Liana Lareau has been co-leading the Innovative Genomics Institute effort to monitor mutations in the SARS-CoV-2 virus in the local community. The team’s rapid sequencing turnaround is crucial for tracking the spread of new variants, including the more infectious B.1.1.7 recently detected in two cases at Berkeley.
Read MoreNew CRISPR-based COVID-19 test uses smartphone cameras to spot virus RNA
Fletcher Lab, in collaboration with the Gladstone Institutes and Doudna Lab, have developed a new CRISPR-based COVID-19 diagnostic test that, with the help of a smartphone camera, can provide a positive or negative result in 15 to 30 minutes. This test also gives an estimate of viral load, or the number of virus particles in a sample, which can help doctors monitor the progression of a COVID-19 infection and estimate how contagious a patient might be.
Read MoreFDA authorizes first rapid at-home covid-19 test, made by Lucira
On Tuesday the first rapid, at-home COVID-19 test was authorized by the Food and Drug Administration. The test, made by BioE-founded company Lucira, uses their loop mediated amplification reaction process, a molecular nucleic acid amplification technology, in a battery-powered standalone unit. Rather than detecting antibodies, their test is designed to detect whether an individual is shedding the coronavirus that causes COVID-19. The company was developing this technology for an at-home influenza test before rapidly pivoting to address COVID-19. Lucira is led by Debkishore Mitra (PhD 2013), who founded the company with John Waldeisen (PhD 2012).
Read MoreBerkeley Bioengineers impress in Biomedical Engineering Virtual Summer Internship
Four bioengineering undergraduates spent the summer working online to increase PPE compliance in universities as part of the first ever Biomedical Engineering Virtual Summer Internship hosted through the Cleveland Clinic and Case Western Reserve University. Check out each student’s idea pitch in the videos below:
Read MoreGauss-Cellex rapid at-home Covid-19 antigen test
Gauss Surgical – a company launched by MTM alumnus Siddarth Satish – has partnered with Cellex to develop the first-ever rapid, at-home COVID-19 antigen test and mobile app.
Read MoreLareau receives LGR Seed Funding award for COVID-19 Project
Prof Liana Lareau and collaborator Stacia Wyman have received one of ten Excellence in Research Awards from the Laboratory for Genomics Research (LGR), a collaboration between UC Berkeley/UCSF (IGI) and GlaxoSmithKline. Their project will explore rapid, low-cost, high-throughput viral and metagenome sequencing of COVID-19 patient samples for outbreak surveillance.
Read MoreHead-Gordon leads COVID-19 Drug-Delivery Simulation Repository
The Molecular Sciences Software Institute (MolSSI) has launched an open-source website that will allow biomolecular scientists from around the world to share computer-aided drug-testing simulations targeting the protein at the center of COVID-19. Under the leadership of MolSSI co-director Teresa Head-Gordon, a MolSSI team started work on the COVID-19 website in April, after scores of scientists began discussing ways to share simulation modeling data they had on the coronavirus.
Read More